- An orbital is the probability map for the electron(s).
- -the electron is somewhere inside the orbital surface 90% of the time. The
- electron does not move around on the surface.
- -the electron is somewhere inside the orbital surface 90% of the time. The
- electron does not move around on the surface.
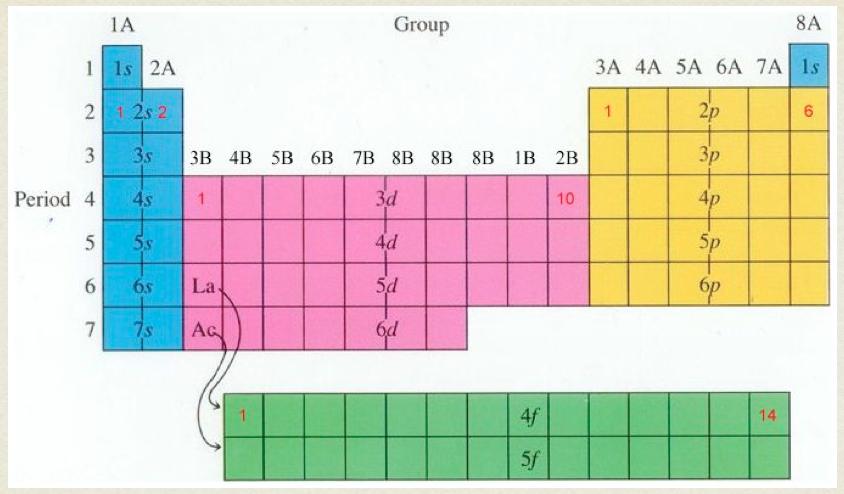
- Orbital labeling rules
- The number tells the principal energy level
- The letter tells the shapes
s orbital
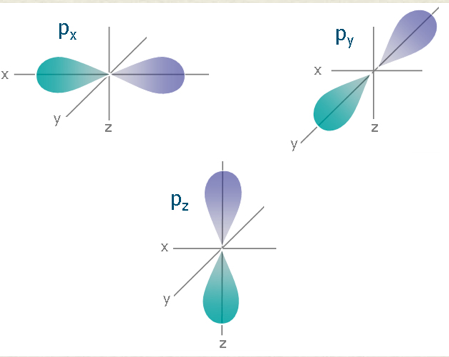
p orbital
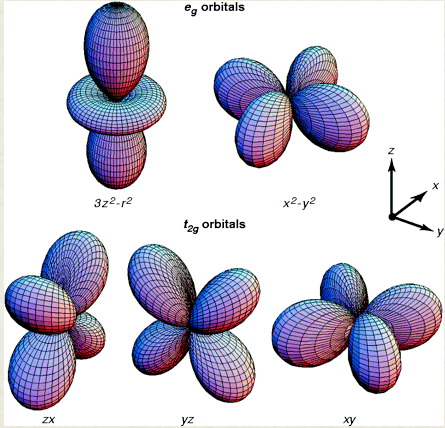
d orbital
Paul Exclusion Principle
- An atomic orbital can hold a maximum of two electron and those two electron must have opposite spins.
- we can represent the spin of the electron with these arrows