Type 1
1. the cation is always named first and the anion named second
Example:
Now you try:
- Cation will have the same name as the element
- Anion will have an ide ending.
- There some exceptions with polyatomic ions
Example:
- Na+ Cl-
- Cation name - Sodium
- Anion name - Chloride not Chlorine
- Correct type 1 naming: Sodium Chloride
Now you try:
- MgS
- Did you get Magnesium Sulfide? Good!
Type 2
- This set deals more with the transition metals.
- Transition metals can have more than one charge.
- These compounds will have systematic names identifying what type of charge is on the metal atom
- Metal name(roman numeral) Anion name
- Example: Iron(II) Oxide
- Convert the name of the compound to the chemical formula.
- Iron (III) Oxide
- First write the symbol
- Fe= Iron and it has a charge of +3
- O=oxide and has a charge of -2
- Fe +3 O -2
- Fe2O3
- Convert the name of the compound to the chemical formula.
- Metal name(roman numeral) Anion name
- Now you try: FeCl2
- Did you get Iron(II) Chloride? Great!
Type 3
- These Compounds contain 2 or more nonmetals that are covalently bonded.
- The first element in the formula is named first. and the full element name is used.
- The second element is named as though it was an anion
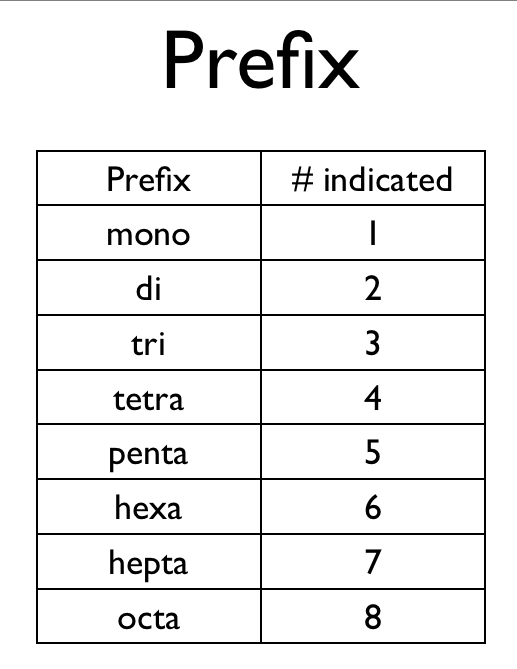
- Prefixes are used to denote the number of atoms
- The pre-fix mono is never used for naming the first element
- example CO is called carbon monoxide
- not monocarbon monoxide
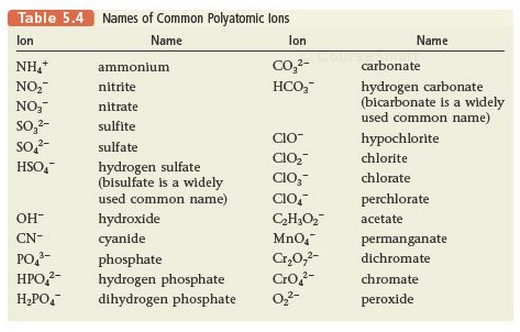
Polyatomic Ions
Here is a list of polyatomic ions.
- These ions contain more than one atoms.
- Example SO4 -2 Sulfate
- These polyatomic ions are used as discrete units
- Example Na+ and SO4 -2
- Combined the compound that would would be formed is Na2SO4
- You can use either type 1 or type II naming schemes. Remember, the nomenclature is based on the cation
- Now you try: What is the chemical formula for this compound? Iron(III) Nitrate
- Here in this video you will find how to do the naming for ionic compounds in type 1, 2, and with polyatomic ions.
Acids
- Acids can be initially described as molecules that will release one or more H+ when attached to an anion.
- HF instead of hydrogen Fluoride
- drop the ide and it will become ic acid and add hydro to the prefix
- hydrofluoric acid
Naming Acids with Oxygen:
- HX
- X being any anion
- Determine the anion:
- Determine the name:
- If the name has ATE ending change it to IC
- If the name has an ITE ending change it to OUS
- also add at the end the word acid
- Drop hydro- prefix
- HNO3
- Determine the anion: NO3
- Determine name: Nitrate
- Since Nitrate has an -ate ending the name will become Nitric Acid.
Here is a video better explaining how to name acids: