Dalton's Atomic Theory

Atoms are the smallest unit of matter. Each atom consists of negatively charged electrons, positively charged protons, and neutral neutrons. Each atom represented by an element's chemical symbol. For example, a lithium atom would be represented like this Li. The number of protons determines the element that the atom will be. You change the number of protons, you change the element all together.
Dalton's Atomic Theory:
1. Each atom present is represented by its element symbol
2. The number of each type of atom is indicated by a subscript written to the right of the element symbol
3. When only one atom of a given type is present, the subscript 1 is not written.
Dalton's Atomic Theory:
- Most natural materials are mixtures of pure substances
- Pure substances are either elements or combination of elements called compound
- Law of constant composition :
- A given compound will always contain the same proportion of elements
1. Each atom present is represented by its element symbol
2. The number of each type of atom is indicated by a subscript written to the right of the element symbol
3. When only one atom of a given type is present, the subscript 1 is not written.
Isotopes
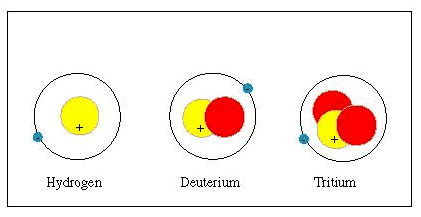
If you remember from the "How to" section of Elements, you learned there is an electron for every proton and to find the number of neutrons you have to subtract the number of protons from the atomic mass. In an ideal situation, the number of neutrons should come out nicely. Some elements have isotopes. Isotopes are atoms of the same element that have a variance in the number of neutrons. Since the hydrogen atom does not have any neutrons, there are special hydrogens that contain one neutron (deuterium) or two neutrons (tritium).
Allotropes
- Non metal solids are more varied than those of metals
- Allotropes- different forms of a given element
- Example- Carbon(C)
- 1. Diamond
- 2. graphite
- 3. Bucky ball( buckminster fullerene)