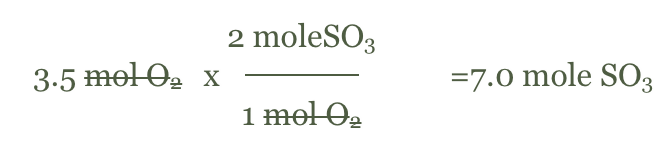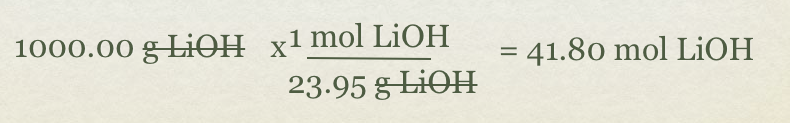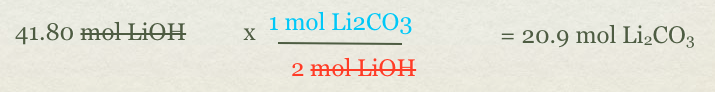Stoichiometry
The process of using chemical equation to calculate the relative masses of reactant and products.
Mole to Mole Relationship
- All mole to mole relationship comes from balanced equations
- Balanced 2SO2(g) + O2(g) -> 2SO3(g)
- If we have 3.5 moles of O2(g) how many moles of SO3(g) can be formed?
- Write the given and used the balanced equation to find the mole to mole ratio.
Mole to mole is the HEART of stoichiometry! So make sure you understand how to do this. Here is a video further explaining mole to mole conversions. It is important you remember how to do conversions from the previous chapters such as grams to moles or moles to grams.
Gram to Mole
- LiOH(s) + CO2(g)->Li2CO3(s) +H2O(l)
- Problem how many moles of Li2CO3(s) will be formed with 1000g of LiOH?
- Step 1 write you know and what you are looking for.
- given:1000g LiOH
- Target: ?? mol Li2CO3(s)
- Step 2 balance equation
- 2 LiOH(s) + CO2(g) -> Li2CO3(s) + H2O(l)
- This is important because these coefficient will be needed for the mole to mole conversion.
- Step 3 set up the problem (convert gram to mol of the same compound)
- We have grams of LiOH but need to go to mol of Li2CO3(s)
- In order to go from one compound to another compound you need moles.
- First, convert grams of LiOH to mol of LiOH using molar mass of the compound
- Step 4 Convert Mol of a compound to mole of another compound
- We have mole of LiOH but need to go to mol of Li2CO3(s)
- In order to go from one compound to another compound you need moles based on the balanced equation.
Now if the question asked for grams of Li2CO3, we would take the answer 20.9 mol Li2CO3 and convert it to grams. See? That isn't too bad!
Percent Yield
Here is a video explaining this concept. How much do I have and how much I can make?
Step by Step!
- Identify the given and target compound
- Balance the equation for the reaction
- Set up the problem (convert to moles if necessary)
- Use the mole ratio(s) to calculate the number of moles of the desired compound
- Convert to grams of the desired compound if necessary