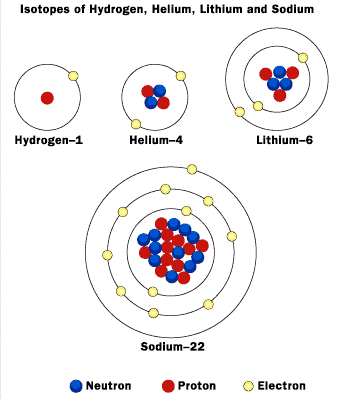
Bohr's Model
Bohr's model works well for Hydrogen and Helium, but other experiment showed that his atomic model does not work for other elements.
Wave Mechanical Model
- By the mid 1920s, people discovered that the Bohr model was incorrect.
- Louis Victor de Broglie(France) and Erwin Schrodinger( Austria) suggested that light have both wave and particle characteristic.
- They believed the electron might also exhibit both of these characteristic
- Bohr’s model, electrons was assumed to move in a circular orbits.
- Wave Mechanic on the other hand can suggest the probability of the location of the electron.
- Probability map or orbitals describes electron in the lowest possible energy state.
- The more intense the color of a given dot, the more likely the electron can be found
- The model gives no information about when the electron occupies a certain point in space or how it moves.